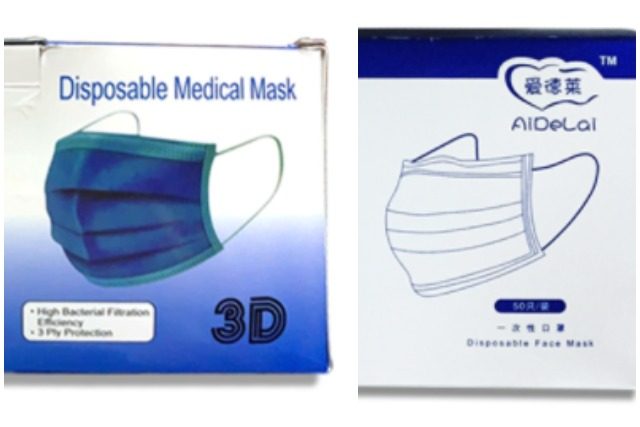
The Food and Drug Administration has cautioned the public against the use and purchase of two brands of face masks that have been widely available in the market since the start of the novel coronavirus pandemic last year.
In two separate advisories on Friday, FDA Director Eric Domingo, also an undersecretary of the Department of Health, announced that the face masks with the names “3D Disposable Medical Mask” and “AiDeLai” have not yet been “notified” and “certified.”
Domingo noted that the FDA cannot evaluate them for quality and safety without certifications.
“The FDA verified through post-marketing surveillance that the above mentioned medical device product is not notified and no corresponding Product Notification Certificate has been issued,” Domingo said.
“Pursuant to the Republic Act No. 9711, otherwise known as the ‘Food and Drug Administration Act of 2009,’ the manufacture, importation, exportation, sale, offering for sale, distribution, transfer and non-consumer use, promotion, advertising or sponsorship of health products without the proper authorization is prohibited,” he added.
In line with this, all establishments were ask to halt distribution, advertisement and sale of these products until the appropriate Product Notification Certificates are issued or they may face face regulatory actions and sanctions.
Domingo also urged local authorities to help in ensuring these products would be pulled out and not sold in the market.
The Bureau of Customs was also directed to prevent enter of these uncertified devices in the country.
AiDeLai as a counterfeit brand?
The AiDeLai brand might be popular among consumers in the Philippines.
However, a report from Taiwan News stated that it is actually a fraudulent manufacturer.
The report, published June 2020, stated that boxes of the product indicated they were manufactured by a certain “Taibei Rongyi Textile Co.” (台北市融億紡織有限公司) and that the company is in Taiwan.
This company does not exist in the East Asian island.
The Taiwan News reporter ran a fact check and found that a company called “Rongyi Textile Co.” is located in China’s Jiangsu province.
Aside from the questionable whereabouts, the article also noted that the boxes of such protective gear contained factual errors.
Taipei, Taiwan’s capital, was misspelled as “Taibei” and claimed that Taiwan is part of China.
Taiwan, officially called Republic of China, is a separate country with its own government from its neighbor.
Are the advisories too late?
Both the “3D” masks and the “AiDeLai” masks have already been sold in online platforms and commercial establishments since the outbreak of the deadly virus which causes COVID-19 back in early 2020.
Filipinos particularly cited AiDeLai in the comments section and questioned the FDA’s advisory.
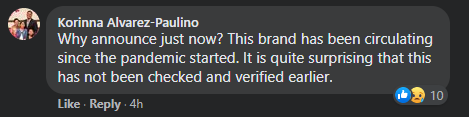
Other users also shared photos of their recent purchases of the face mask brand and that they found nothing wrong with them.
We bought this from Savemore Sta. Rosa Laguna. We used it today, nothing unusual about it. 😐 pic.twitter.com/gtEcMBCzbK
— Raymond Cerezo (@raymondcerezo) January 15, 2021
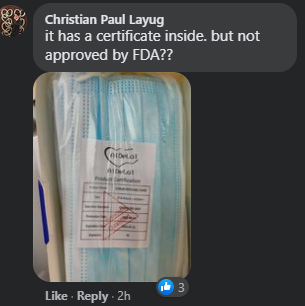
Some Facebook users, meanwhile, urged the FDA to disclose a list of brands of face masks that the organization had approved and evaluated.
Health organizations have no recommended brands for face masks and shields.
What they discourage is the wearing face masks with exhalation valves as they don’t block the transmissions of potentially infective respiratory droplets.
The health authorities reminded the public that face mask should also completely cover one’s nose and mouth but still allows one to breathe.